Health
New findings reveal success in Ebola immunisation
A partnership of some of the world’s leading health organisations has announced publication of data from a Phase 1 study of a preventive Ebola vaccine regimen. The results, first published in the The Journal of the American Medical Association on the vaccine regimen, suggest that the regimen produced immune response in health volunteers.
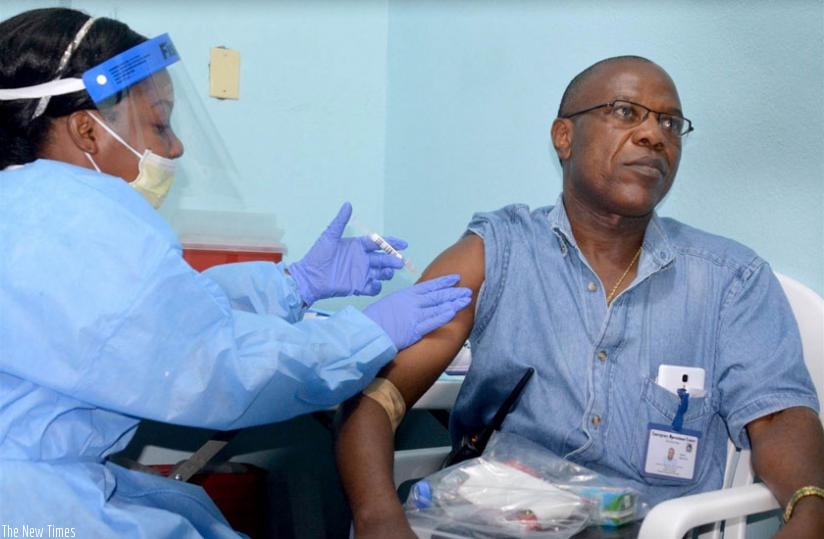
Doctor Francis Kateh, right, from Redemption Hospital volunteers to receive a trial vaccine against Ebola at Redemption Hospital on the outskirts of Monrovia, Liberia. /(Net photo)
